Pancreatic cancer cell atlas highlights why many promising treatments fail
Published on 30/03/2026
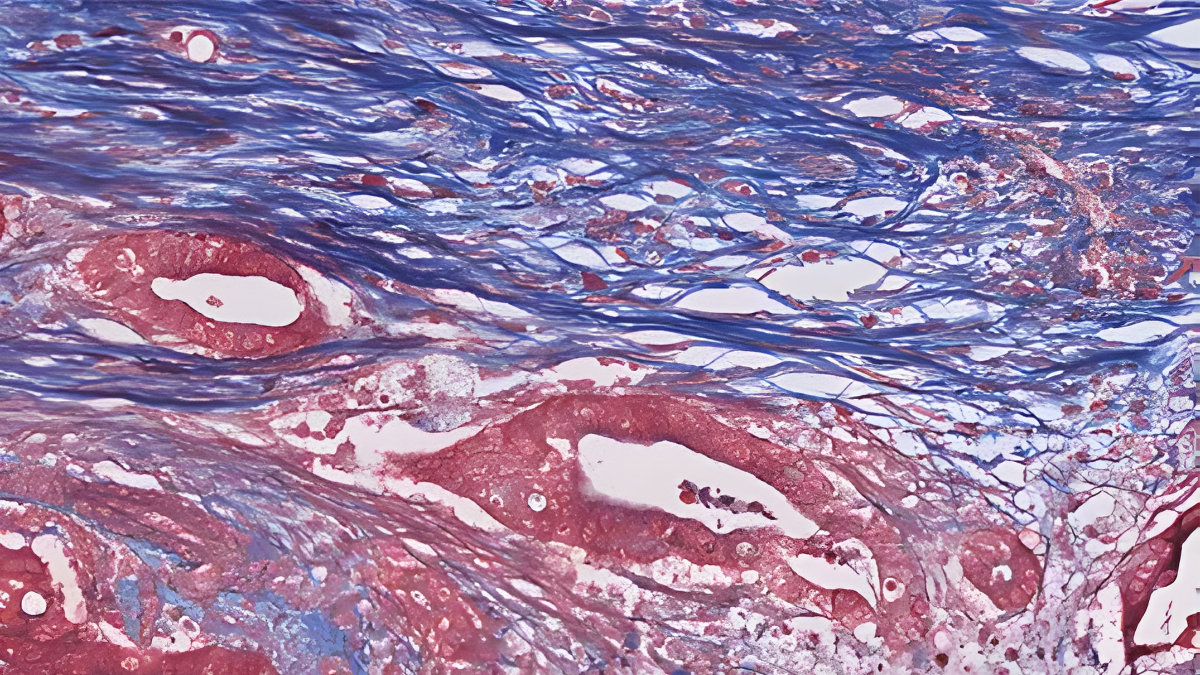
The most detailed atlas of tumour cells from the deadliest form of pancreatic cancer has been developed by an international team of researchers, and the findings uncover how tumour cells change their behaviour depending on their surroundings—and why many promising treatments fail in standard lab tests.
The research is a joint collaborative effort led by the University Hospitals Birmingham (UHB) and the University of Birmingham in the UK, and the pharmaceutical company Bristol Myers Squibb.
Published in Cell Reports, their findings describe the most detailed spatial map to date of pancreatic ductal adenocarcinoma (PDAC), an extremely aggressive disease that accounts for 90% of all pancreatic cancers.
In the study, researchers analysed tumour samples from 39 untreated pancreatic cancer patients, using cutting-edge spatial transcriptomics technologies that allow scientists to see which genes are active in cells and exactly where those cells are located in the tumour.
This approach generated a massive dataset—hundreds of thousands of spatial measurements and more than half a million individual cells—creating a comprehensive “atlas” of pancreatic cancer tissue.
Pancreatic cancer remains one of the hardest cancers to treat, with few effective therapies and a five-year survival rate in the single digits. A major reason is the disease’s complexity: cancer cells are embedded in a dense, hostile tissue environment filled with scar-like material, low oxygen, and supportive cells that help tumours survive.
The study highlights several important findings:
Cancer cell identity depends on location. The study confirmed known pancreatic cancer subtypes, commonly called classical and basal-like, but showed that these identities are strongly shaped by the tumour’s local environment. In particular, aggressive basal-like cancer cells were consistently found in regions with low oxygen and dense fibrotic tissue (thick, scar-like tissue that can surround and protect tumours, making them harder to treat).
Researchers discovered that an “intermediate” tumour subtype is not just a mix of known states, but a distinct cancer cell identity. This finding clarifies long-standing confusion in pancreatic cancer classification. They also identified a previously underappreciated group of highly proliferative cancer cells, marked by intense cell-division activity. Although relatively rare, these cells may disproportionately drive tumour growth.
The team used CRISPR gene-editing screens (a tool that allows scientists to make precise changes to DNA) in cancer cells grown under realistic conditions, such as low oxygen or inside living tumours. This revealed genetic weaknesses that are invisible in standard laboratory cultures, and may explain why many drug targets that look promising in the lab fail in patients.
Furthermore, common lab models miss key disease features. Widely used mouse and cell-line models captured some aspects of human pancreatic cancer but often failed to reproduce the most aggressive tumour states and their surrounding environments. This highlights the need for better preclinical testing strategies.
Dr Shivan Sivakumar, Consultant Medical Oncologist at UHB and co-senior author from the University of Birmingham, said: “This spatial atlas is expected to serve as a foundational resource for the research community and may accelerate the development of treatments for a disease that has long resisted progress.
“By integrating spatial biology with functional genetic screening, we have created a roadmap for discovering therapies that target pancreatic cancer more effectively—especially combination treatments designed to disrupt both cancer cells and the environments that protect them.
“The findings suggest that environmental factors play a much greater role in tumour cell development in the pancreas, and identifies new intermediate tumour subtypes and highly proliferative cancer cells which together provide a more vivid picture of this deadly cancer.”
Dr. Konstantinos Mavrakis, Executive Director and Head of Discovery Biosciences Oncology at Bristol Myers Squibb and co-senior author of the publication emphasises the importance of scientific collaborations: “This study underscores how important it is to use real-world patient data to better understand the underlying causal human biology of a specific cancer type such as pancreatic cancer.”


